|
Later on in 1913, complete agreement of Rutherford’s proposal was found with the experimental results of Geiger and Marsden. Some particles even scattered in the backward direction making 180° angle.įrom this experiment, Lord Rutherford first proposed that scattering of α-particles through large angles is only possible if all the positive charges and the mass of the atom are concentrated at the center covering only a very small space around it. In this experiment, they noticed that some alpha particles went straight through the foil and some particles were scattered making small angles and some went making the angle more than 90°. This experiment is known as the famous Rutherford’s alpha deflection experiment. In 1909, scientists Geiger and Marsden, at the suggestion of Ernest Rutherford, performed a deflection experiment of alpha-particles having an energy of 7.68 MeV emitted from the radioactive polonium, on a 6 x l0 -7 m thick gold foil. Each atom contained the same number of positive and negative charges. Within this positively charged matter, negatively charged electrons were randomly distributed. As a result, he discovered the emission of a subatomic particle which, in 1919, he called the "hydrogen atom" but, in 1920, he more accurately named the proton.Throughout the nineteenth century, scientists had the idea that each atom was composed of positive charges and these charges were distributed all over the atom. He performed the first artificially induced nuclear reaction in 1917 in experiments where nitrogen nuclei were bombarded with alpha particles. Rutherford's explanation, which he published in May 1911, was that the scattering was caused by a hard, dense core at the center of the atom–the nucleus. In the now well-known experiment, alpha particles were observed to scatter backwards from a gold foil. An alpha particle consists of two protons and two neutrons, which is actually a helium nucleus. The nucleus emits an alpha particle and energy. The Figure below shows what happens during alpha decay. What causes alpha decay?Īlpha decay occurs when a nucleus is unstable because it has too many protons. In 1909 the physicist Ernest Rutherford directed an experiment at the University of Manchester in England to measure small deflection angles recently observed when alpha particles–tiny positively charged bodies given off by radioactive elements–were beamed through a thin gold foil. When did Rutherford directed a stream of alpha? Alpha particles come from the decay of the heaviest radioactive elements, such as uranium, radium and polonium. What is an alpha particle made of?Īlpha particles (α) are positively charged and made up of two protons and two neutrons from the atom's nucleus. In 1899 Ernest Rutherford studied the absorption of radioactivity by thin sheets of metal foil and found two components: alpha (a) radiation, which is absorbed by a few thousandths of a centimeter of metal foil, and beta (b) radiation, which can pass through 100 times as much foil before it was absorbed.  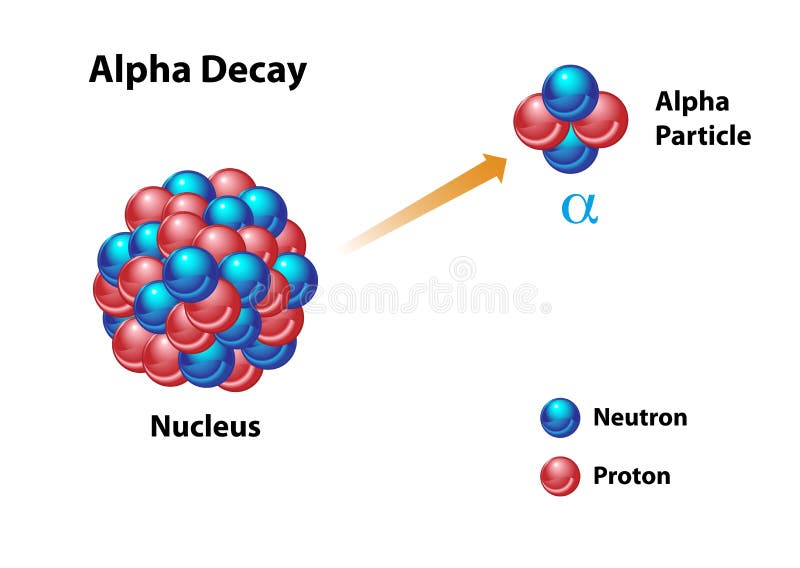
How did Rutherford discovered alpha and beta rays? He discovered that there are two types of radiation, alpha and beta particles, coming from uranium. Where did Ernest Rutherford get the alpha particles?Įrnest Rutherford is known for his pioneering studies of radioactivity and the atom. Because the nucleus is unstable a piece of it is ejected, allowing the nucleus to reach a more stable state. How alpha particles are produced?Īn alpha particle is produced by the alpha decay of a radioactive nucleus. They collected α particles in a sealed glass tube, compressed them, and passed an electric spark through. How did Rutherford make alpha particles?įor this work Rutherford recruited Thomas Royds (1884–1955), who had earned his Physics Honours degree in 1906. Geiger later informed Rutherford that they could see the occasional deflection by greater than 90 degrees of one alpha particle for every 8000 particles traversing the gold foil. They used radium as the source of the alpha-particle beam to bombard a thin foil of gold.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
